A clinically validated, gene expression-based test guiding personalized treatment decisions for patients with Multiple Myeloma.
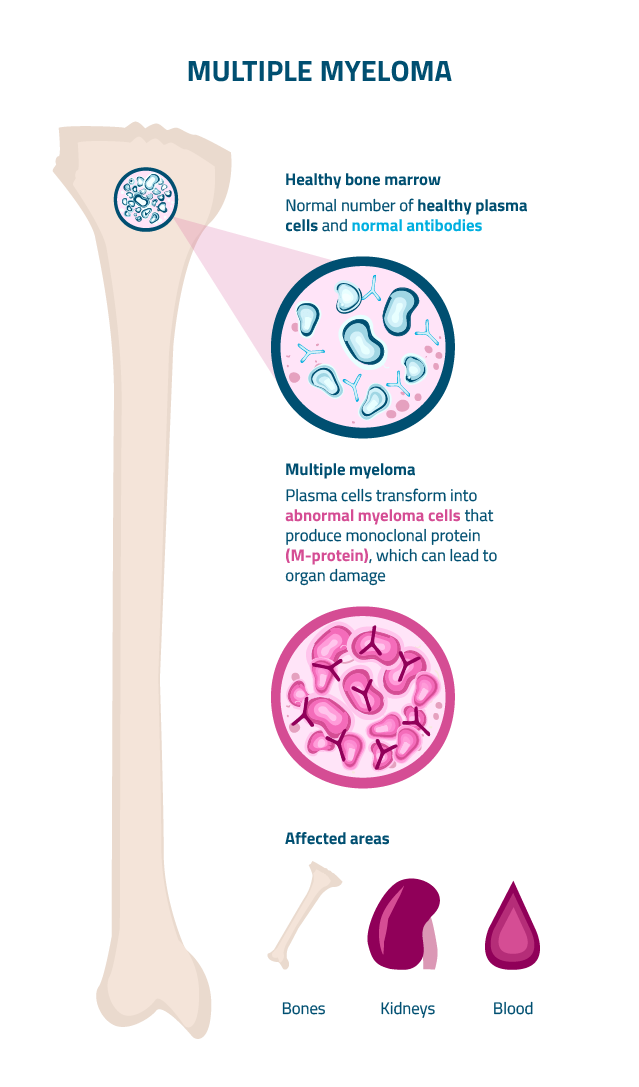
Multiple Myeloma (MM) is the second most common blood cancer. It starts in plasma cells, which are white blood cells found mainly in your bone marrow. The disease can behave differently in each patient, ranging from slow-growing forms to more aggressive types.
MM develops when malignant plasma cells multiply uncontrollably in the bone marrow, leading to an excessive buildup of abnormal myeloma cells. These cancerous cells crowd out healthy blood cells and produce large amounts of abnormal proteins called monoclonal proteins (M-proteins). Over time, this can lead to serious complications, commonly referred to as CRAB symptoms, which include:

Elevated serum calcium levels from increased bone resorption

Kidney impairment from free light chain deposition and hypercalcemia

Decreased red blood cell production due to bone marrow infiltration

Osteolytic lesions, pathological fractures, and bone pain due to increased osteoclast activity
While multiple myeloma remains a serious and currently incurable condition, advances in molecular diagnostics, such as with gene expression profiling test, are improving our ability to predict disease behavior and personalize treatment, helping to extend survival and improve quality of life.
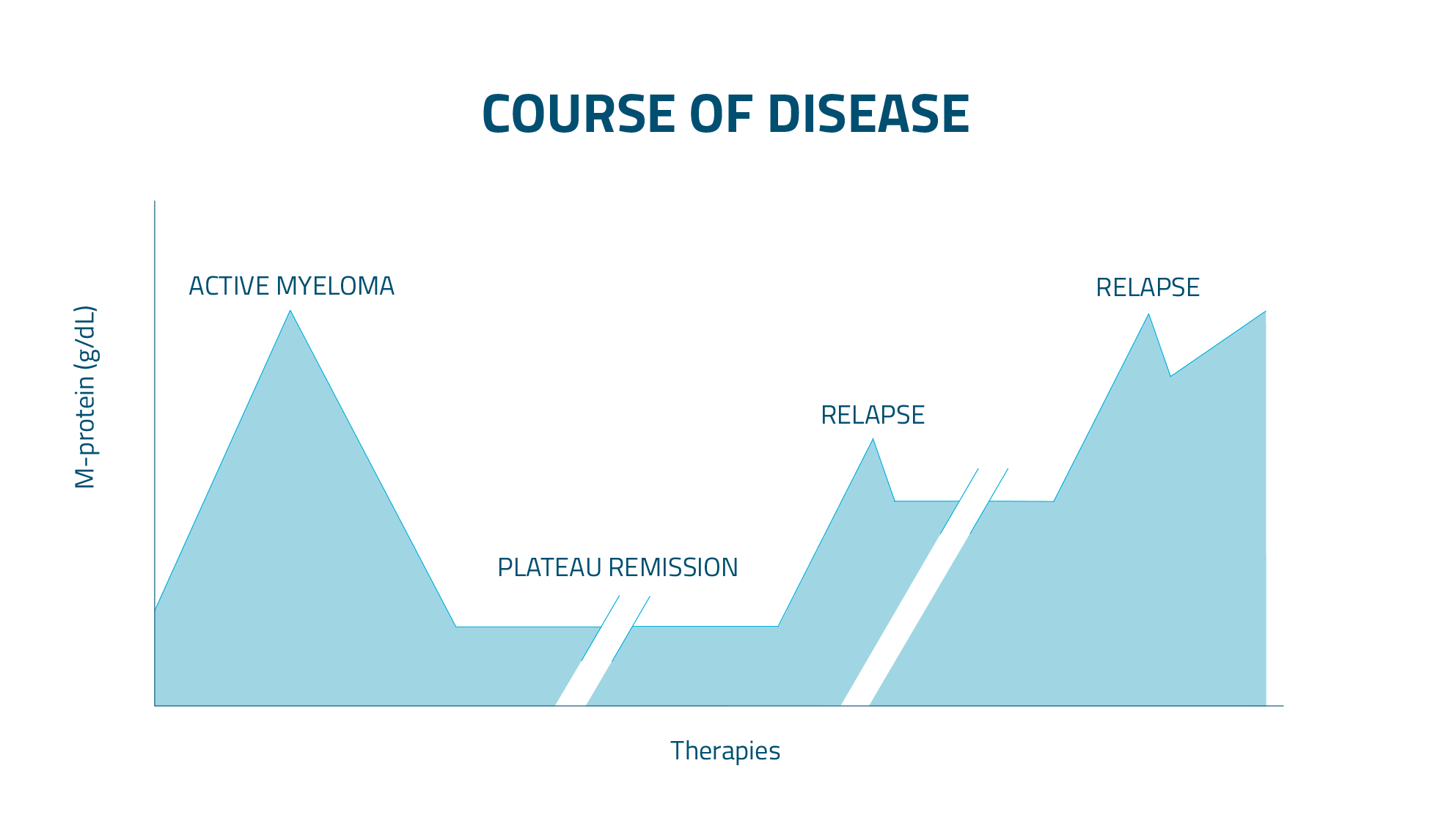
Myeloma is a cancer that typically follows a relapsing-remitting course. During active periods, patients experience symptoms or complications that require treatment. Treatment can lead to remission periods when symptoms are minimal or absent. However, most patients will experience disease recurrence and with each recurrence, treatments often become less effective. That’s why choosing the right first-line therapy is especially important since it offers the best chance to shape long-term outcomes.

Multiple myeloma demonstrates significant heterogeneity in disease progression and treatment response. Some patients experience less aggressive disease with long survival, while others face aggressive progression despite intensive therapy.
Early and accurate risk assessment at diagnosis is crucial for guiding treatment decisions in multiple myeloma.
SKY92 offers standardized risk classification based on tumor biology at diagnosis and relapse, helping clinicians make confident, personalized treatment decisions. By revealing the disease’s biology early, they guide more targeted strategies potentially improving outcomes for patients.
SKY92 is a molecular diagnostic test developed by SkylineDx designed specifically for patients with Multiple Myeloma (MM) — both newly diagnosed and those with relapsed or refractory disease.
It analyses the expression of 92 genes, known as the SKY92 signature, in myeloma cells to uncover prognostic information not available through standard diagnostic methods. It has a binary outcome:
Standard Risk
indicates a more favourable prognosis, with a lower risk of rapid disease progression and better expected survival outcomes
High Risk
indicates a higher likelihood of aggressive, progressive disease and reduced overall survival
SKY92 empowers clinicians with more precise risk stratification, enabling informed treatment decisions that can improve patient outcomes

SKY92 is a molecular diagnostic test developed by SkylineDx designed specifically for patients with Multiple Myeloma (MM), both newly diagnosed and those with relapsed or refractory disease.
It analyses the expression of 92 genes, known as the SKY92 signature, in myeloma cells to uncover prognostic information not available through standard diagnostic methods.
It has a binary outcome:
Standard Risk
indicates a more favourable prognosis, with a lower risk of rapid disease progression and better expected survival outcomes
High Risk
indicates a higher likelihood of aggressive, progressive disease and reduced overall survival
*SKY92 is marketed in the USA as an LDT and in Europe under the name MMprofiler CE IVD assay kit, with CE IVD and UKCA mark.
SKY92 empowers clinicians with more precise risk stratification, enabling informed treatment decisions that can improve patient outcomes
3500+ patients validated across 21 clinical cohorts
Endorsed by several myeloma guidelines
The test is analytically validated with a standardized, quality-control process to ensure reliability and accuracy
Presented in 60+ scientific publications at leading international hematology and oncology conferences
CE-IVD marked in the EU, UKCA-marked in the UK, and LDT certified in the USA
Sample Collection
Bone marrow aspirate is collected during standard diagnostic procedure.
Cell Isolation
CD138+ plasma cells are isolated using magnetic bead separation.
RNA Extraction
Total RNA is extracted and quality-controlled for analysis.
Gene Expression Analysis
Expression of 92 genes is measured using microarray technology
Risk Classification
Proprietary algorithm stratifies patient into High or Standard Risk
Identifying a patient’s risk level at diagnosis allows doctors to tailor treatment strategies more effectively:
If the risk is identified as…
Standard Risk
SKY92 Standard Risk patients generally have a stable, slow-growing disease and may do well with conventional therapies and could benefit from less intensive treatment, helping preserve options for future relapses.
If the risk is identified as…
High Risk
SKY92 High Risk patients are generally rapidly progressing, genetically unstable disease (classified as high-risk) typically need more intensive first-line treatment. This may include:
Close monitoring is essential from the start, and these patients may also be eligible for clinical trials specifically designed for high-risk disease.
Diagnosis and initial treatment of transplant-eligible high-risk myeloma patients
Patients tested
Across 17 clinical cohorts
Published Studies
In peer-reviewed journals demonstrating the clinical utility and prognostic value
Abstracts
at leading international hematology and oncology conferences
Academic institutions
worldwide have helped to
develop the test
Contact SkylineDx to learn more about implementing SKY92 in your practice, accessing educational resources, or requesting information about reimbursement and logistics.
While traditional tests like FISH (Fluorescence In Situ Hybridization) identify certain chromosomal abnormalities, SKY92 detects gene expression patterns that reveal biological behavior missed by standard methods. In fact, SKY92 identifies 10–15% of high-risk patients not captured by clinical markers like FISH.
SKY92 is suitable for patients with newly diagnosed or relapsed/refractory Multiple Myeloma, especially when a more precise risk assessment could impact treatment planning. It is typically ordered by hematologists or oncologists managing MM care.
SKY92 has been analytically and clinically validated in multiple international studies.
SKY92 supports personalized medicine by:
The SKY92 test is completed in approximately 4 working days in the laboratory. In a clinical setting, accounting for sample logistics and processing workflows, results are typically delivered in less than 3 weeks from the time of sample collection